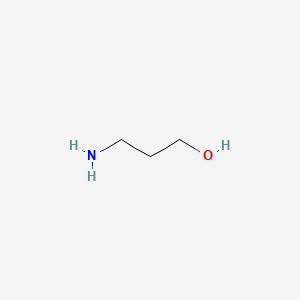
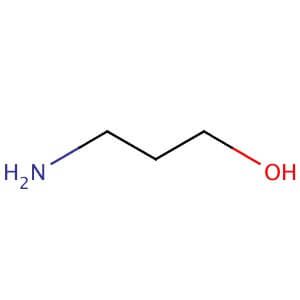
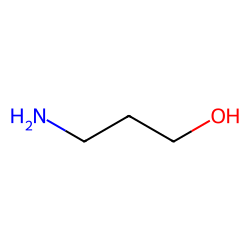
USN Open Archive: Density and Viscosity Correlations for Aqueous 3-Amino-1- propanol and Monoethanol Amine Mixtures

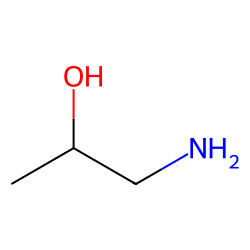
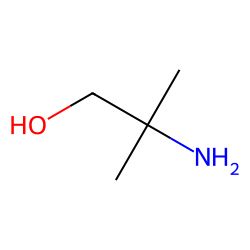
1-amino-3-methoxy-2-propanol - 93372-65-7, C4H11NO2, density, melting point, boiling point, structural formula, synthesis

Viscosity Measurement and Correlation of Unloaded and CO2-Loaded 3-Amino-1- propanol Solution | Journal of Chemical & Engineering Data

Experimental Study of Some Thermodynamic Properties of Binary Mixtures Containing 3-Amino-1-propanol, 2-Aminoethanol, and 1-Butanol at Temperatures of 293.15–333.15 K to Model the Excess Molar Volumes Using the PFP Theory | Journal of

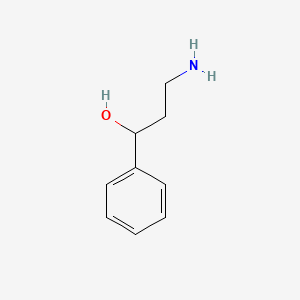
1-amino-3-phenyl-2-propanol - C9H13NO, density, melting point, boiling point, structural formula, synthesis